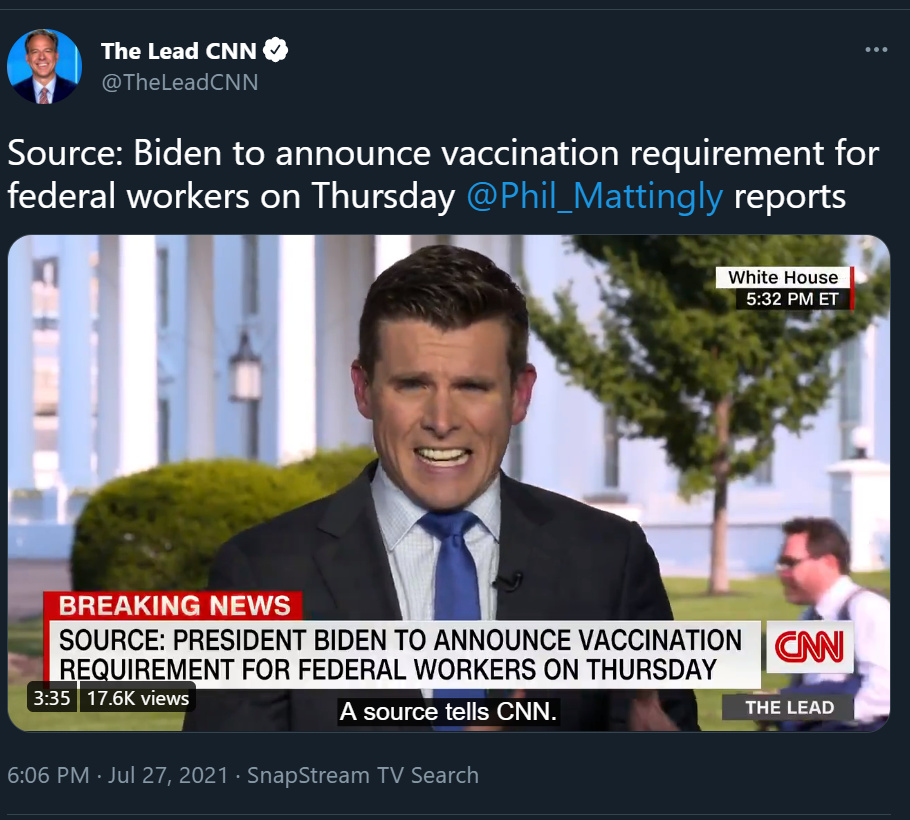
Science magazine has a nice write up. The FDA has had the application for ages -“ Pfizer and Moderna both received an EUA in December 2020; J&J’s came in February. Based on the real-world data they have collected since then, Pfizer applied to FDA for full approval in early May, and Moderna on 1 June. J&J is expected to follow soon
…
On 16 July, FDA accepted Pfizer’s application “under priority review”—meaning it will move faster than during standard reviews, which typically take at least 10 months; the agency now has until January 2022 to review the materials”
I wish they would get a move on. This is a bit more important than approving an Alzheimer’s drug that doesn’t actually work. This quote from the article sums up my thinking, although I am neither an infectious disease doc nor in public health
“I think it’s fair to say that any number of us who are clinical infectious disease doctors and in public health are frankly a little surprised at how long the process is taking,” says William Schaffner, professor of infectious diseases at Vanderbilt University Medical Center.
“I want [FDA] to be careful. I also want them to move it along,” Schaffner says. “Frankly, I’d like them to work on the weekends. The people who are vaccinating are working on the weekends. The virus is working on the weekends.”